The current focus of the Pham Lab is to develop novel approaches to deliver therapeutics that will halt or reverse inflammation and joint degeneration in preclinical models of rheumatoid arthritis, osteoarthritis, and other inflammatory conditions, with the ultimate goal of translating these findings to the clinic. These projects represent a team-science, interdisciplinary approach to arthritis research, combining the Pham lab expertise in basic mechanisms underpinning these rheumatic/arthritic/inflammatory conditions with innovative bioengineering advances in nanomedicine and regenerative medicine pioneered by expert collaborators in the Departments of Orthopedics, Biomedical Engineering, Biology, the Center of Regenerative Medicine, and the Genome Engineering & Stem Cell Center at McDonnell Genome Institute.
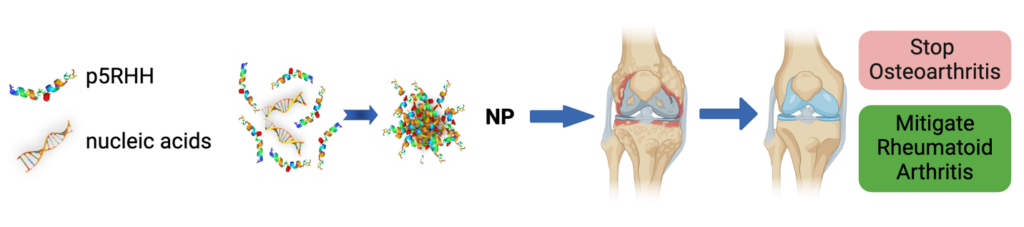
We employ a novel peptide-siRNA nanocomplex targeting the NF-kB pathway to mitigate inflammation in murine models of rheumatoid arthritis and post-traumatic osteoarthritis. We have shown that peptide-NF-kB p65 siRNA nanocomplex suppresses experimental rheumatoid arthritis and mitigates several important early events post-joint injury and pain. We have also shown that the silencing of periostin, a matricellular secretory matrix protein, ameliorates post-traumatic osteoarthritis. In addition, we have used this flexible peptide platform to successfully deliver mRNA to overexpress anabolic factors in injured cartilage/chondrocytes. These findings may lead to the development of a first-in-class disease-modifying nanotherapeutic approach to prevent or mitigate post-traumatic osteoarthritis.
https://doi.org/10.1073/pnas.1608245113; https://medicine.wustl.edu/news/nanoparticle-injections-may-future-osteoarthritis-treatment/; https://doi.org/10.1002/art.41794
We have also collaborated with the Guilak lab to test the ability of a tissue-engineered stem cell-based system with autoregulated cytokine antagonist delivery to mitigate inflammation in a robust murine model of rheumatoid arthritis. The system employs genome-engineered pre-differentiated iPSCs to deliver anti-cytokine therapeutics, the production of which is driven by endogenous levels of inflammatory cytokines. Our data suggest that this “SMART” cell-based delivery of IL-1 receptor antagonist suppresses inflammation, prevents bone erosions and mitigates pain induced by inflammatory arthritis.
https://www.science.org/doi/10.1126/sciadv.abj1414; https://www.nature.com/articles/s41584-021-00705-z; https://medicine.wustl.edu/news/rheumatoid-arthritis-treated-with-implanted-smart-cells-that-release-drug/
More recently, the Pham lab works with a multidisciplinary team of scientists including Farshid Guilak, PhD, (Orthopedic Surgery, Center of Regenerative Medicine), Hua Pan, PhD, (Rheumatology), Lori Setton, PhD, (Biomedical Engineering), Erik Herzog, PhD, (Biology), and Xiaoxia Cui, PhD, (Genome Engineering & Stem Cell Center at McDonnell Genome Institute) to develop treatments that promote tissue regeneration and restore joints in response to the Advanced Research Projects Agency for Health (ARPA-H)’s call for the Novel Innovations for Tissue Regeneration in Osteoarthritis (NITRO) moonshot initiative. The team will leverage the award to create first-in-kind nanoparticle-based platforms for the in vivo delivery of synthetic “smart” gene circuits that home to the joint for long-term regenerative osteoarthritis therapy.
The Pham lab’s research output